 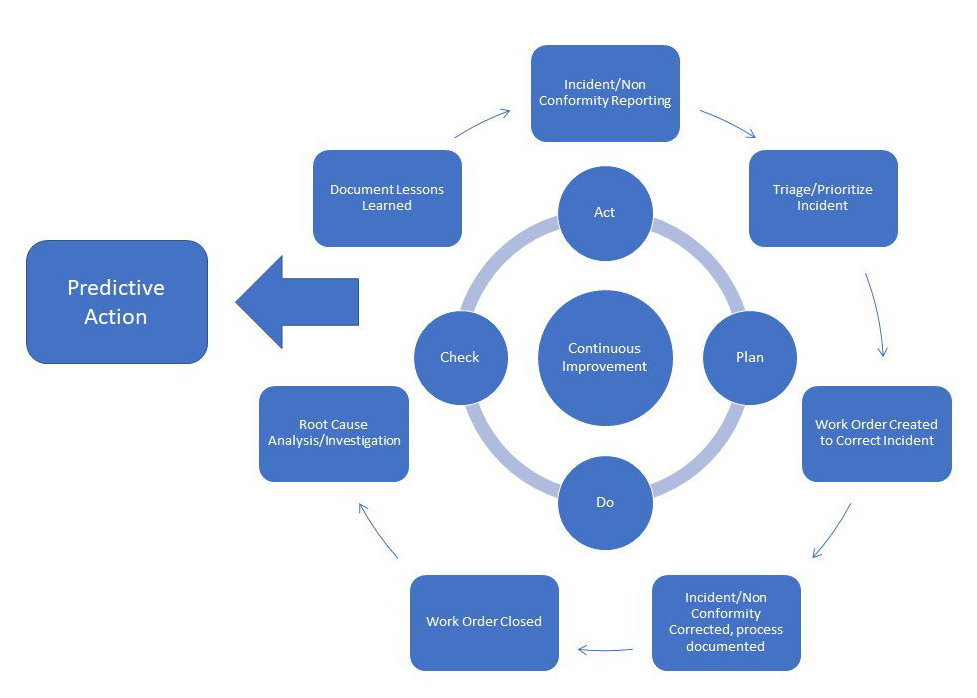
The investigation will analyse and determine the root cause of the problem described, together with any contributing factors. The Quality Management system must have a documented process for investigating any confirmed issues that have been raised: this includes an objective for the actions that will be taken, the procedure to be followed, the personnel that will be responsible for the investigation and any other resources that will need to be accessed in the process. This is the point where the CAPA request should be actioned or denied. This includes assessing the actual risks to the company and or/their customers presented by the failure (particularly any danger to human health and life resulting from it). The problem described and documented above should be evaluated to determine the need for action and the level of action required to solve the issue. To counter this, the CAPA process should only be used for the resolution of systemic problems. 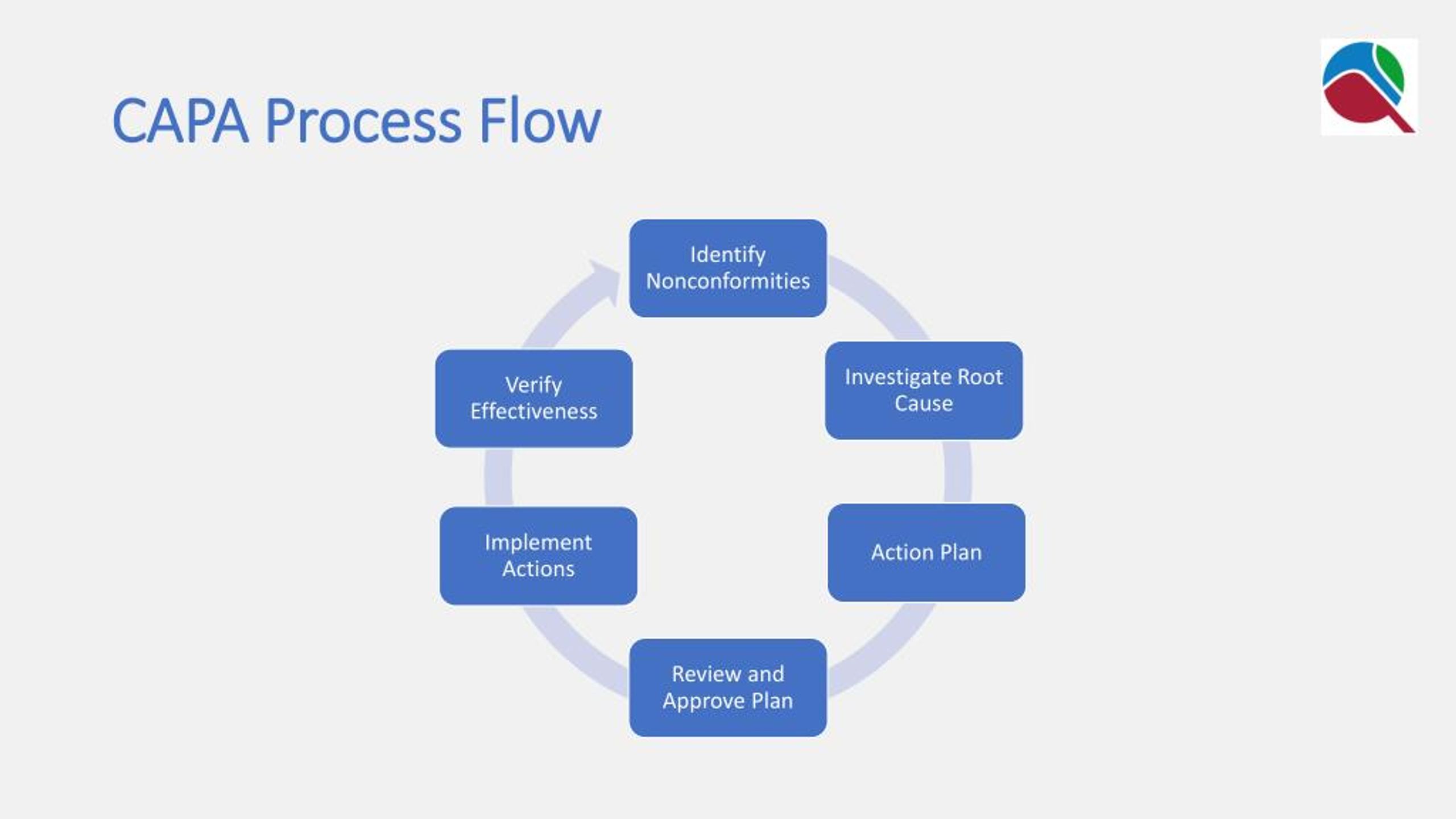 
If it’s used too little, it implies the system is not working properly and nonconformities are remaining unaddressed. If it’s triggered too much (and unnecessarily) it can exhaust time and resource. One of the main issues around CAPA for a business is the frequency with which the process is invoked. The log must include a written description of the problem, the source of the information that initiated the action and a detailed explanation of the problem and the evidence that the problem itself exists. Even if further CAPA is not required or appropriate, there should be a place where the issue can be raised and logged for future auditing and tracking. In the first instance, your Quality Management System must have a way of receiving and logging a problem or potential problem with your end product that someone within or outside the organisation has raised. What are the FDA's requirements for CAPA? Here is what you need to know The 7 steps of CAPA management 1. Such proactive measures include risk assessments, failure modes and effects analysis ( FMEA) as well as audits, management reviews and inspections. This includes not only providing for the swift reporting of existing failures but having proactive measures in place designed to regularly check that systems remain fit for purpose. The purpose of the corrective and preventive action subsystem is to collect information, analyze information, identify and investigate product and quality problems, and take appropriate and effective corrective and/or preventive action to prevent their recurrenceīoth the Corrective Action and Preventative Action you undertake should be part of a seven-step process, that all needs to start with the capturing, recording and defining of the actual or potential problem, non-conformity or incident. They should outline how your company deals with the identification and correction of existing or potential non-conformities in its processes and end products - and how it resolves to prevent them from occurring (or reoccurring) in the future.Īs the supporting FDA material succinctly puts it: Requirements for CAPA management as they apply to the highly regulated world of medical device development can all be found within ISO 13485:2016, the MHRA guidelines, and FDA 21 CFR Part 820.100 But what is CAPA management?ĬAPA refers to two distinct requirements for documented ‘Corrective Action’ and ‘Preventative Action’ procedures that should form part of your med tech Quality Management System. Mastering CAPA management is part of the proactive quality mindset that regulators demand, but it is also an absolute requirement for achieving commercial success in the medical device industry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
