Dart collection of modern human skeletons at the University of Witwatersrand, South Africa, with crania, mandibles, and pelves, were assessed. Materials and Methods: 165 individuals from the 19th to 20th century Inden skeletal series, 252 individuals from the 13th to 14th century Lübeck skeletal series of German ancestry housed at the Department of Historical Anthropology and Human Ecology, the University of Göttingen, Germany, and 161 individuals from the 19th and 20th century of South African African ancestry housed within the Raymond A. Objectives: The focus of this research is to evaluate the sex estimation methods on isolated human materials by applying morphological methods published in various forensic and anthropological literature on different skeletal series. Thus, confinement provides another avenue for manipulating the magnetic properties of this compound. Finally, the antiferromagnetic ordering that characterizes unconfined FeCl 4 is suppressed when the ionic liquid is confined in 5.39-nm pores. Confinement also impedes ion transport as activation energies are elevated when the ionic liquid is placed inside the mesopores. Isothermal pressure-dependent infrared spectroscopy reveals a similar modification to phase transition pressures, with the confined ionic liquid requiring higher pressures to trigger phase transformation than the unconfined system. The confinement-induced reductions in phase transition temperature follow the Gibbs–Thomson equation if a 1.60 nm non-freezable interfacial layer is postulated to exist along the pore wall. 
Isobaric thermal experiments show melting point reductions that depend on the pore diameter of the mesopores. Placing the ionic liquid inside SBA-15 mesoporous silica produces a significant impact on the material’s response to temperature, pressure, and magnetic fields. 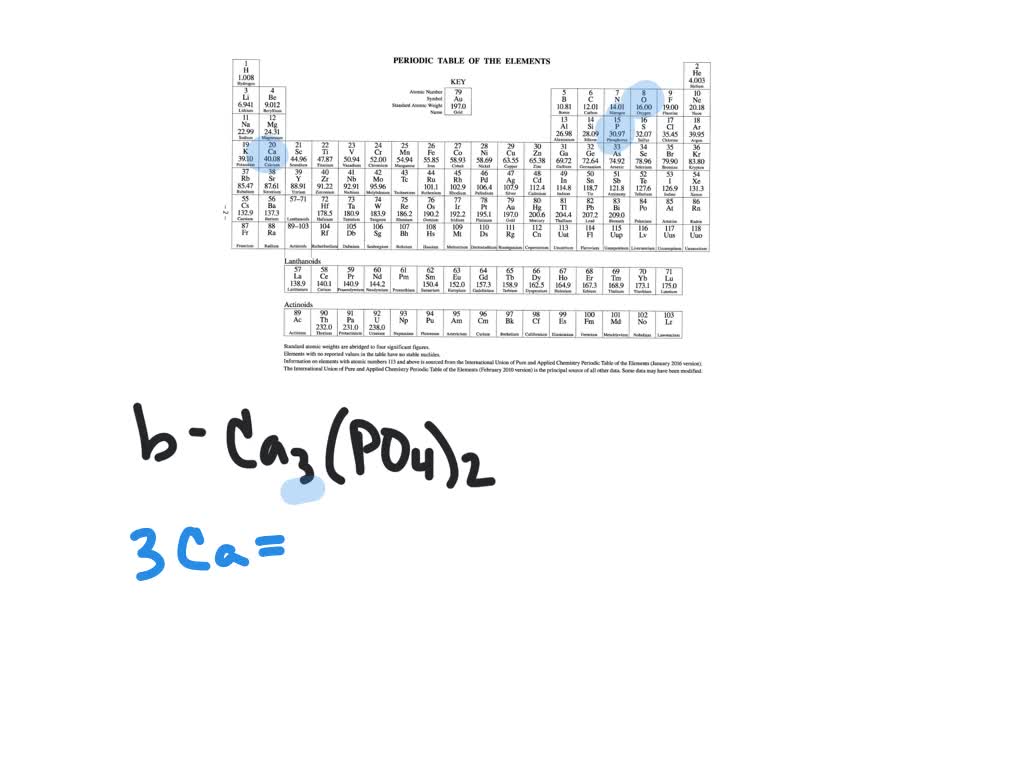
This site explains how to find molar mass.Confinement effects for the magnetoresponsive ionic liquid 1-ethyl-3-methylimidazolium tetrachloroferrate(III), FeCl 4, are explored from thermal, spectroscopic, and magnetic points of view. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Ī common request on this site is to convert grams to moles. 
The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. These relative weights computed from the chemical equation are sometimes called equation weights.įinding molar mass starts with units of grams per mole (g/mol). In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
